I recently had a patient decide to pause treatment for her vector-borne infections because her endometriosis had significantly worsened. The pelvic pain was escalating. Her cycles were becoming more disruptive. Understandably, she wanted to focus on the gynecologic issue first.
But the more I sat with it, the more I wondered whether separating the two may have been a mistake.
I see menstrual cycle changes frequently during antimicrobial treatment. Cycles shorten or lengthen. Bleeding becomes heavier for a month or two. Cramping intensifies temporarily. Sometimes there is mid-cycle spotting that had never occurred before. It can look like hormonal chaos.
It may be die-off related. It may be inflammation altering hormone receptor signaling. It may be immune activation interacting with a tissue that is already exquisitely sensitive to inflammatory shifts.
But what if it is not coincidence?
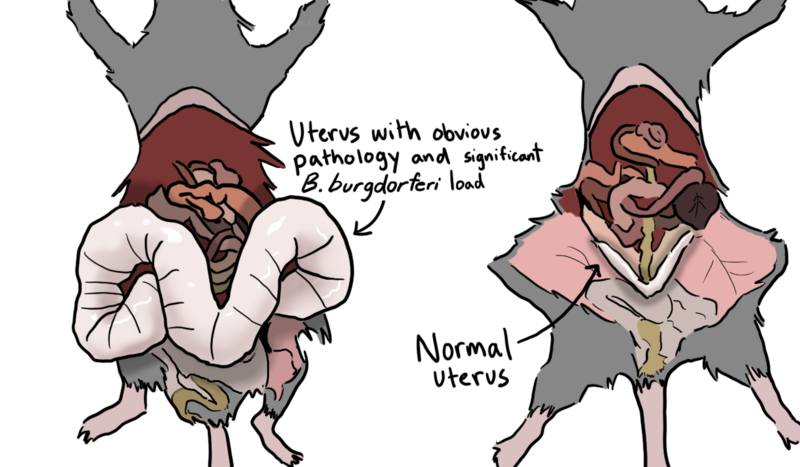
The Uterus Is an Immune Organ
We tend to think of the uterus as a hormonal organ. Estrogen rises, progesterone falls, bleeding begins. Repeat.
But the uterus is deeply immunologic. It cycles through inflammatory and anti-inflammatory states every single month. It must defend against pathogens while tolerating sperm. It must support implantation without overreacting to foreign tissue. It remodels itself continuously.
That is not a passive environment.
When a persistent infection like Borrelia burgdorferi enters the picture, it does not interact with “hormones” in isolation. It interacts with immune cells embedded in reproductive tissue.
Dr. Michal “Mikki” Tal’s research at MIT has begun exploring this intersection. In mouse models, untreated Borrelia infection led to striking uterine pathology — enlargement, cystic changes, structural distortion. Age amplified the severity. What began as an immunology project unexpectedly opened a gynecologic door.
When the team examined human data, they found associations between Lyme disease and higher rates of heavy menstrual bleeding, miscarriage, uterine fibroids, and endometriosis. That does not prove causation. But it challenges the assumption that infection stays in the joints.
For many women, it may not.
Why Treatment Can Temporarily Disrupt the Cycle
When antimicrobial therapy begins, the immune system often shifts. Cytokines rise transiently. Inflammatory mediators fluctuate. Tissue immune signaling changes.
The endometrium is highly responsive to those signals.
Estrogen receptors and progesterone receptors do not operate in isolation. They are influenced by inflammatory pathways. Inflammation can alter receptor sensitivity, local prostaglandin production, vascular tone, and clotting behavior.
So when I see a cycle temporarily worsen during infection treatment, I do not automatically assume hormones are failing.
Sometimes I suspect the immune system is speaking through reproductive tissue.
Backing off infection treatment in that moment may calm short-term symptoms. But if the underlying immune driver remains, the gynecologic manifestations may persist or progress.
Why Women May Be More Vulnerable
Chronic infection-associated syndromes consistently skew female. Chronic Lyme. Long COVID. Many autoimmune conditions.
Women tend to mount stronger antibody responses. They often generate more robust inflammatory signaling. That confers advantages in acute infection survival. It also increases vulnerability to persistent inflammatory states.
If Borrelia persists or dysregulates immune signaling, the reproductive tract — already immune-dynamic — may reflect that imbalance.
This may help explain why endometriosis, fibroids, heavy bleeding, and miscarriage patterns can appear or worsen in the context of infection-associated illness.
Age and Hormonal Transition
Tal’s mouse work suggested older females developed more consistent uterine pathology than younger ones.
Clinically, this resonates.
Perimenopause is an immune transition. Postpartum is an immune transition. Hormonal fluctuations alter immune tone. When infection is present in the background, those transitions may unmask or amplify symptoms.
Many women report that pelvic symptoms intensify not at the beginning of illness, but during hormonal shifts years later.
That may not be random.
The Clinical Implication
If a woman with vector-borne infection develops worsening endometriosis, heavier bleeding, or new pelvic pain, the reflex response is often to separate the issues.
Treat the gynecology. Pause the infection work.
But if infection is contributing to immune dysregulation within reproductive tissue, dividing the problem in two may not address the driver.
This does not mean every case of endometriosis is infectious. It does not mean every heavy period is Lyme-related.
It does mean that in some women, especially those with known vector-borne infections and systemic immune symptoms, the uterus may be part of the same immune story.
A Broader Reframe
For years, women have been told their pelvic pain is stress. Their heavy bleeding is “just hormones.” Their miscarriages are bad luck.
Emerging research suggests the conversation may need to widen.
The uterus is not isolated from the immune system. Hormones do not function independently of inflammation. Infection does not stay neatly compartmentalized.
Sometimes the menstrual cycle is not misbehaving.
It is reporting.

.png)